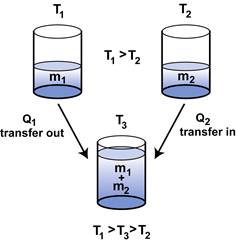
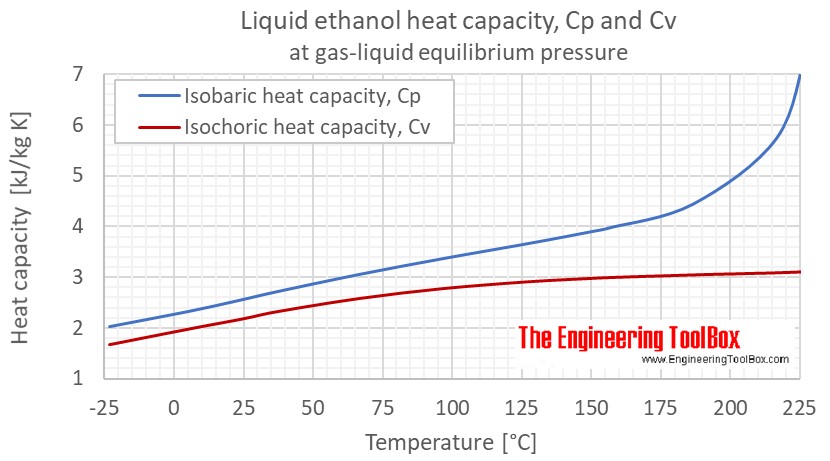
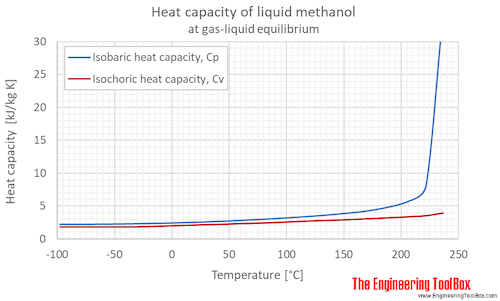
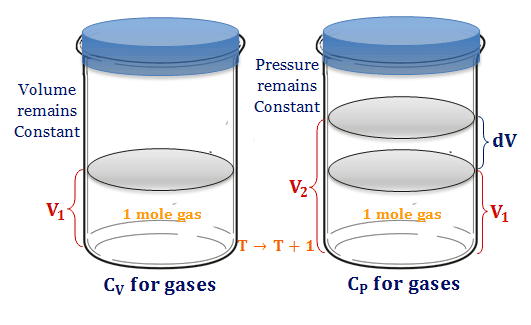
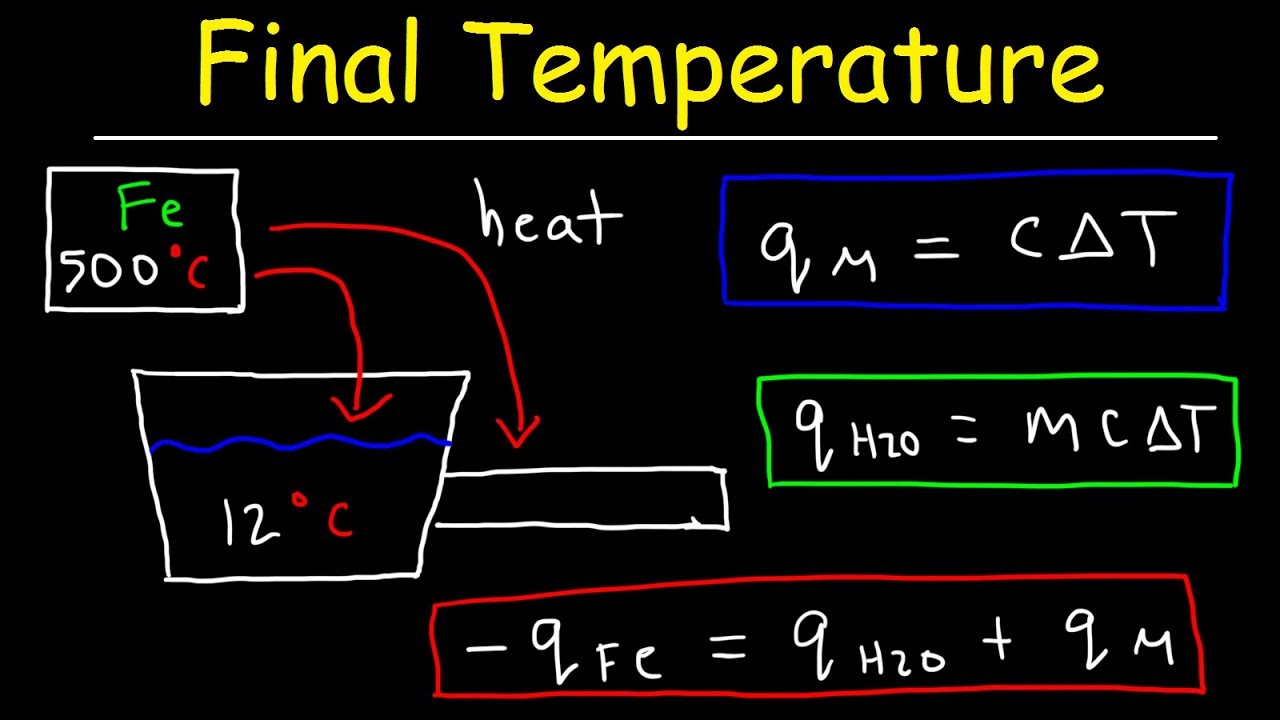
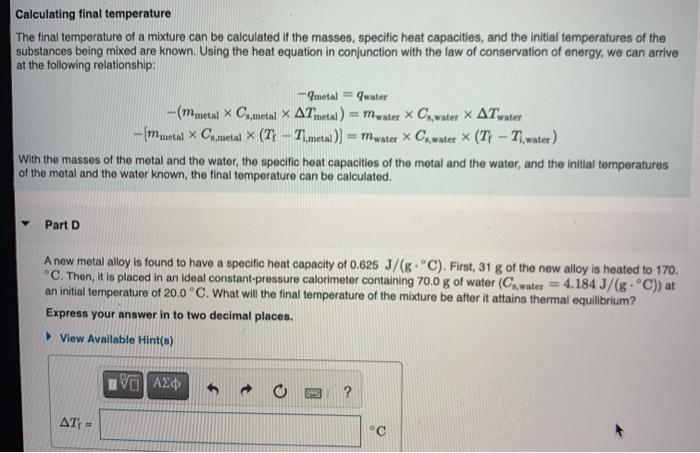
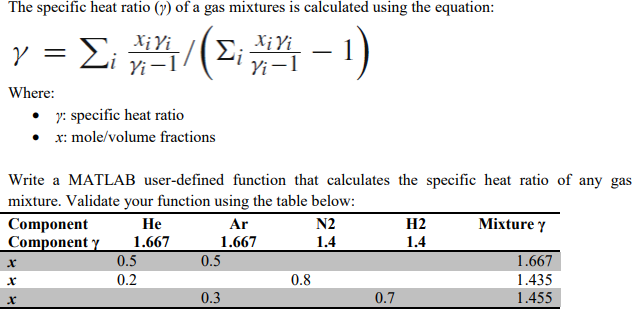
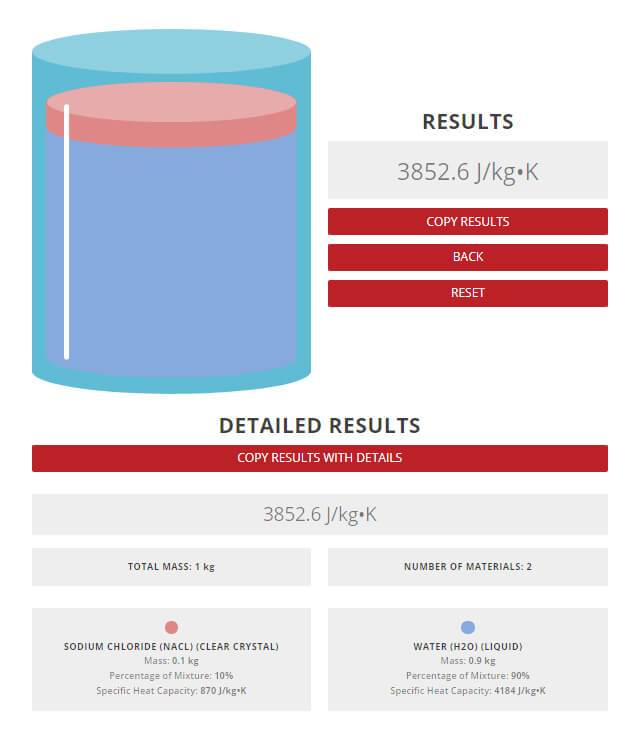
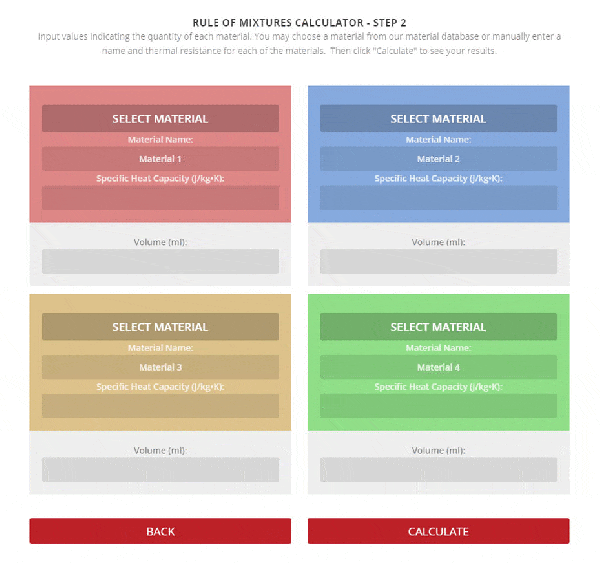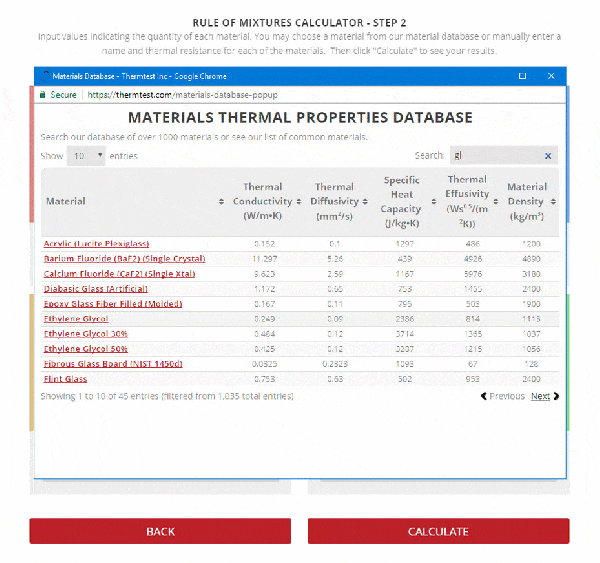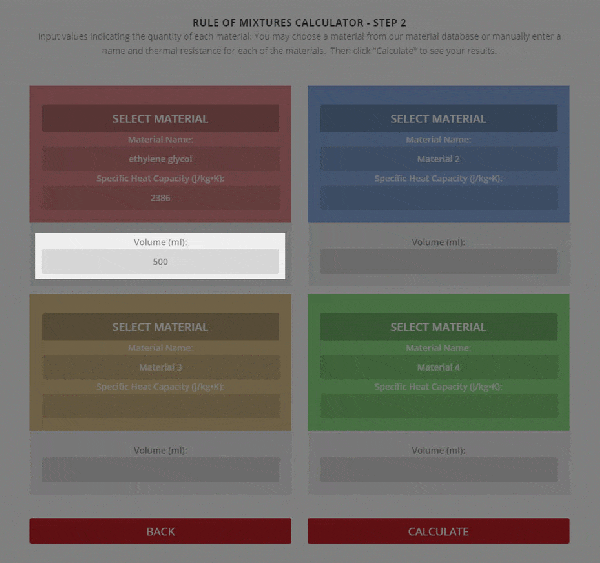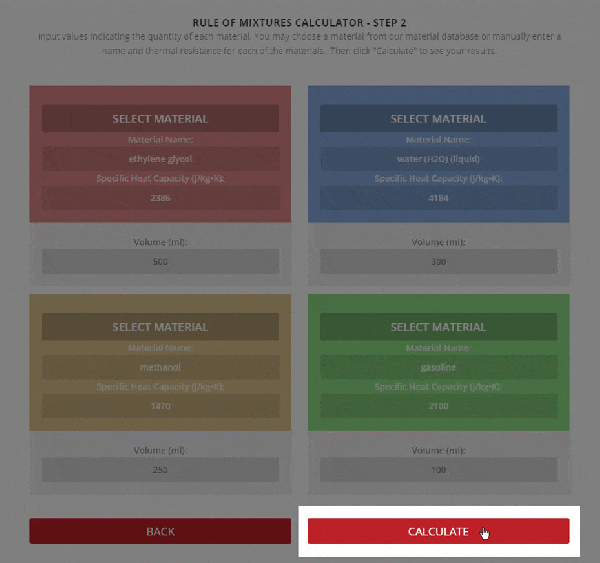
SOLVED: A5. 50-g sample of aluminum pellets (specific heat capacity 0.89 J/'C g) and a 19.00-g sample of iron pellets (specific heat capacity 0.45 J/PC*g) are heated to 100.0 C. The mixture

Thermal fluids with high specific heat capacity through reversible Diels-Alder reactions - ScienceDirect

Find the molar specific heat of mixture at constnat volume. It one mole of a monoatomic gas is mixed with three moles of a diatomic gas.
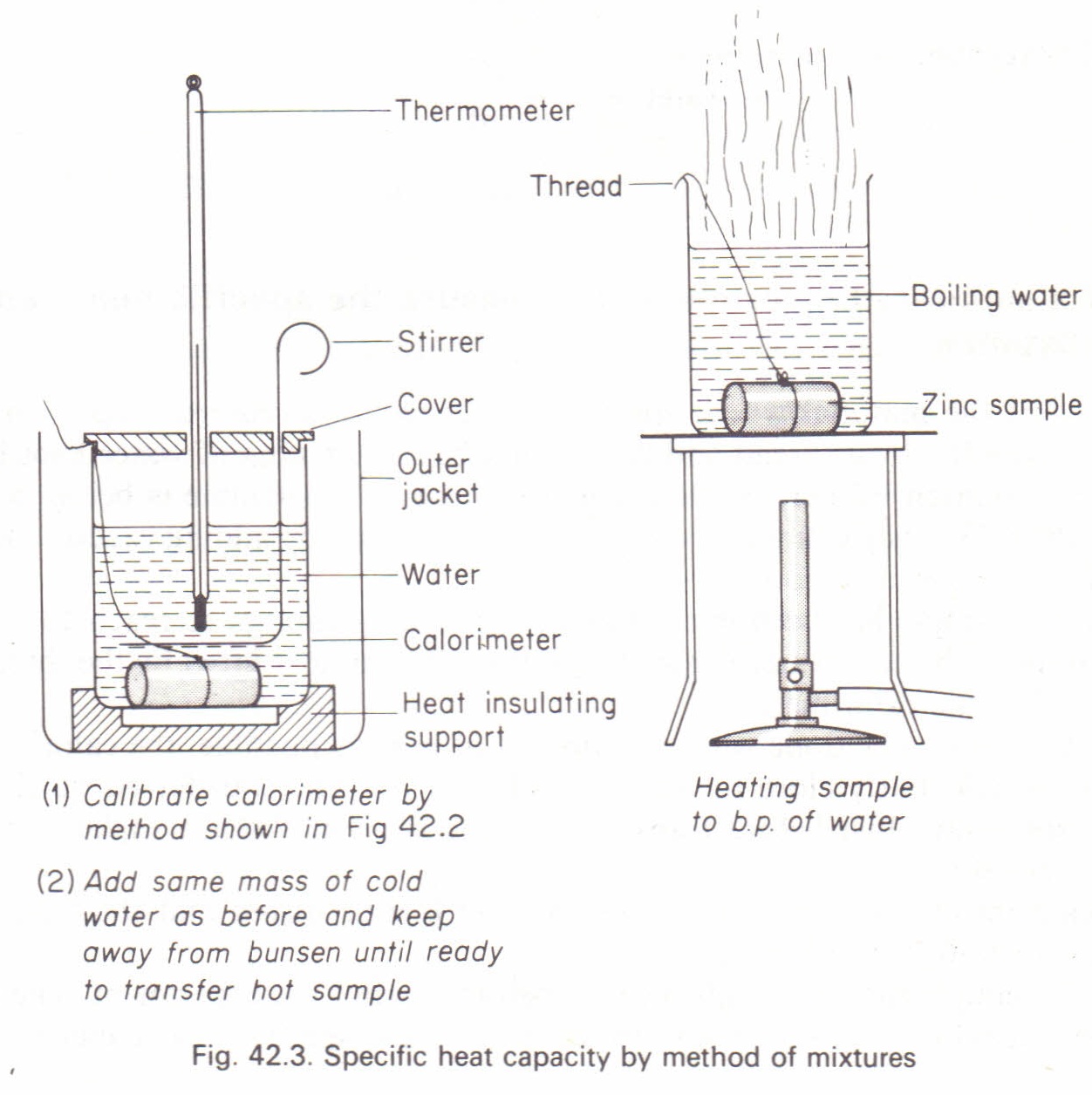
To measure the specific heat capacity by the method of mixtures Physics Homework Help, Physics Assignments and Projects Help, Assignments Tutors online

What is Specific Heat Capacity? | How to Find Specific Heat Capacity - Video & Lesson Transcript | Study.com