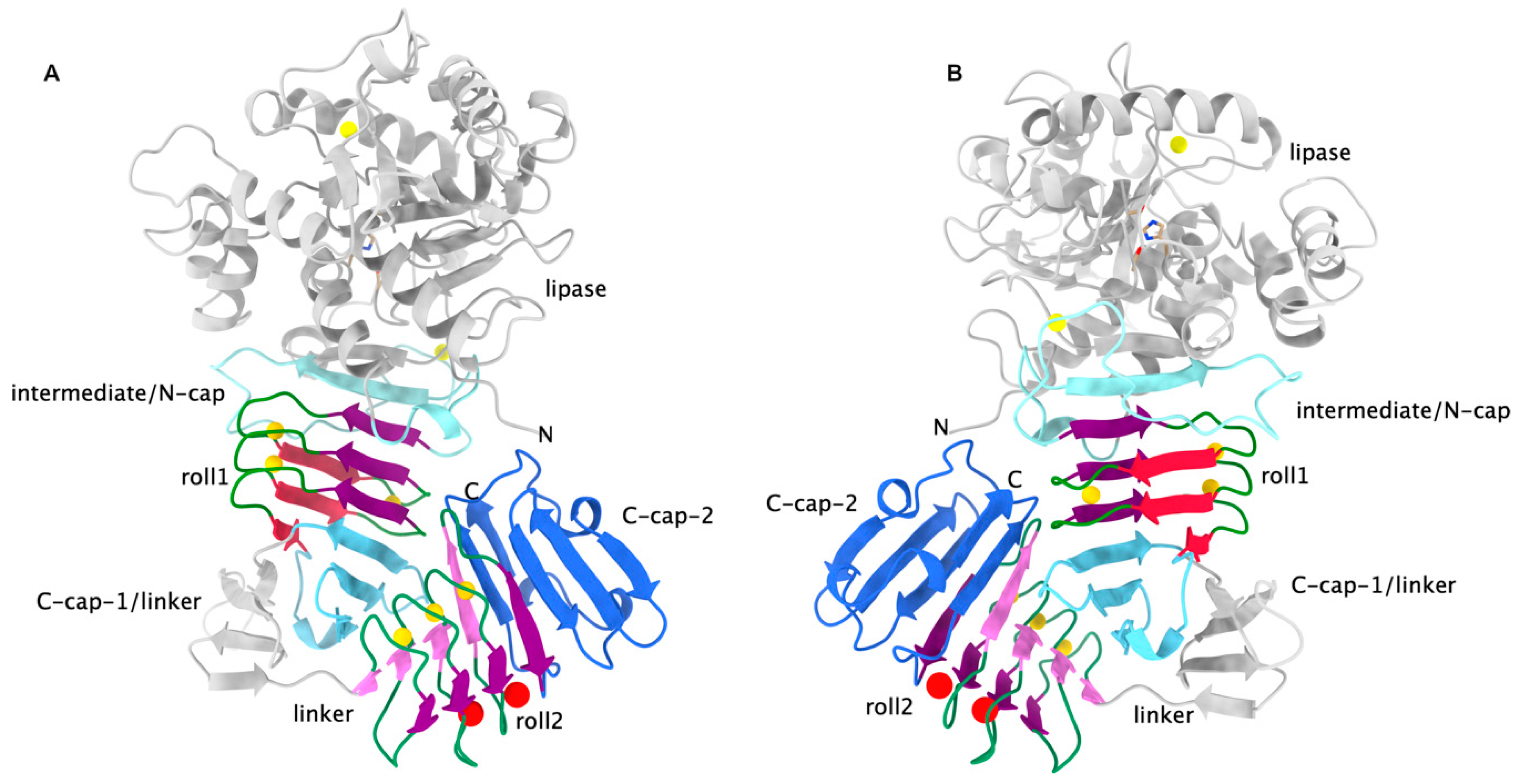
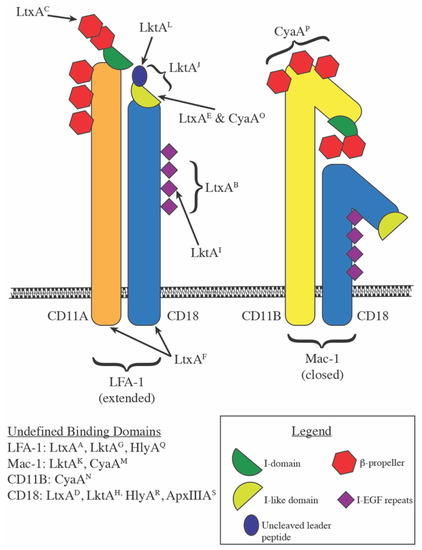
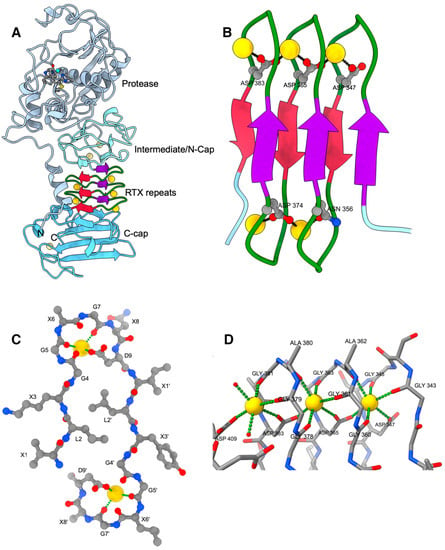
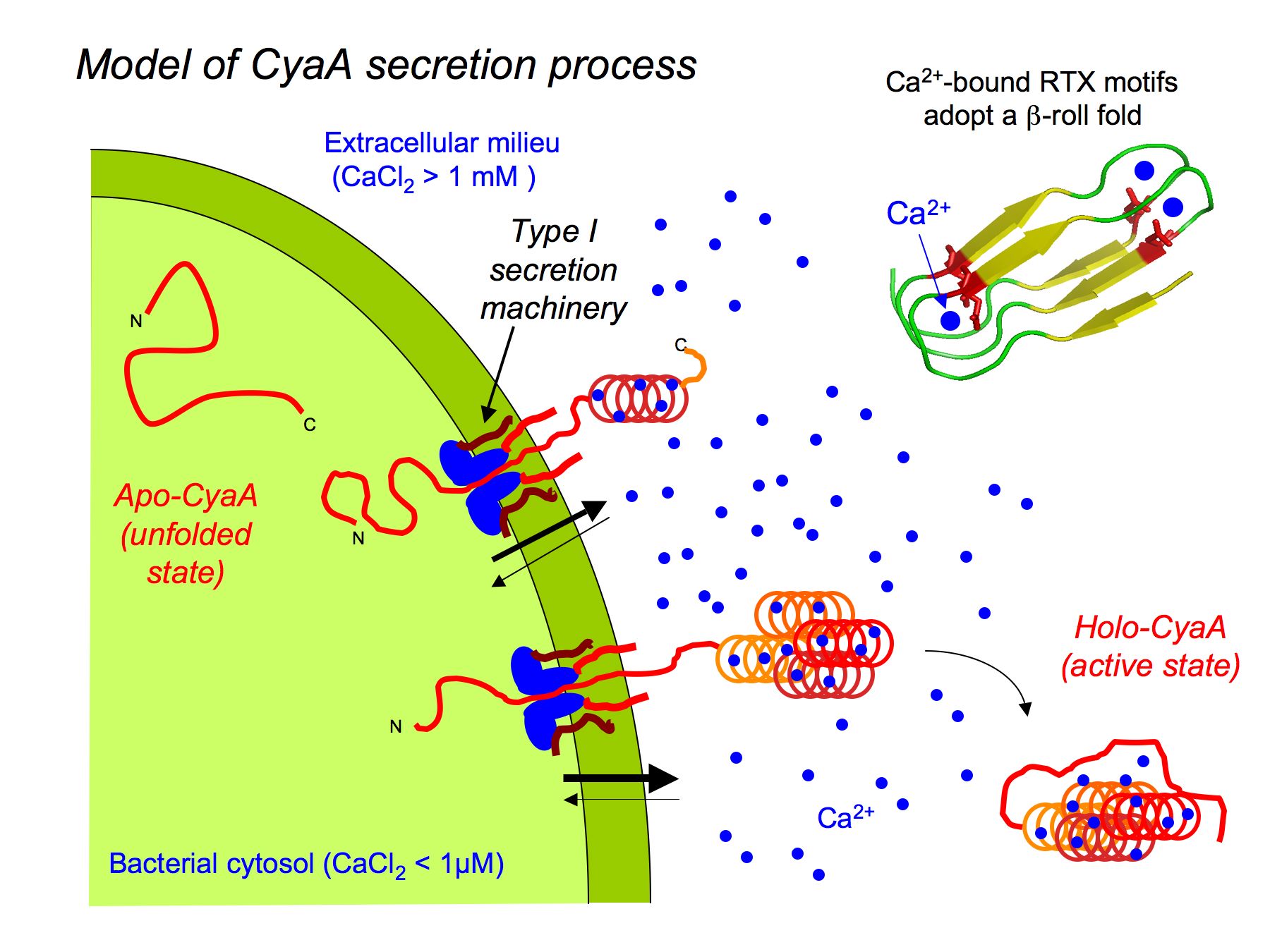
Pore forming activity of the potent RTX-toxin produced by pediatric pathogen Kingella kingae: Characterization and comparison to other RTX-family members - ScienceDirect
Membrane localization of the Repeats-in-Toxin (RTX) Leukotoxin (LtxA) produced by Aggregatibacter actinomycetemcomitans | PLOS ONE

RTX proteins: a highly diverse family secreted by a common mechanism - Linhartová - 2010 - FEMS Microbiology Reviews - Wiley Online Library

Identification of a Vibrio cholerae RTX toxin gene cluster that is tightly linked to the cholera toxin prophage | PNAS
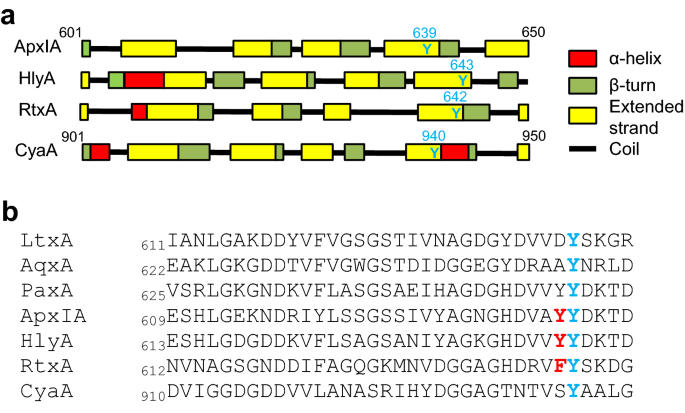
Different roles of conserved tyrosine residues of the acylated domains in folding and activity of RTX toxins | Scientific Reports
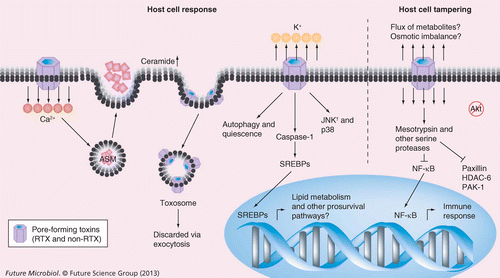
The RTX pore-forming toxin α-hemolysin of uropathogenic Escherichia coli: progress and perspectives | Future Microbiology

2 Schematic organization of HlyA and CyaA RTX toxins. The different... | Download Scientific Diagram

Channel formation by RTX-toxins of pathogenic bacteria: Basis of their biological activity - ScienceDirect

Single Molecule Force Spectroscopy Reveals the Mechanical Design Governing the Efficient Translocation of the Bacterial Toxin Protein RTX | Journal of the American Chemical Society
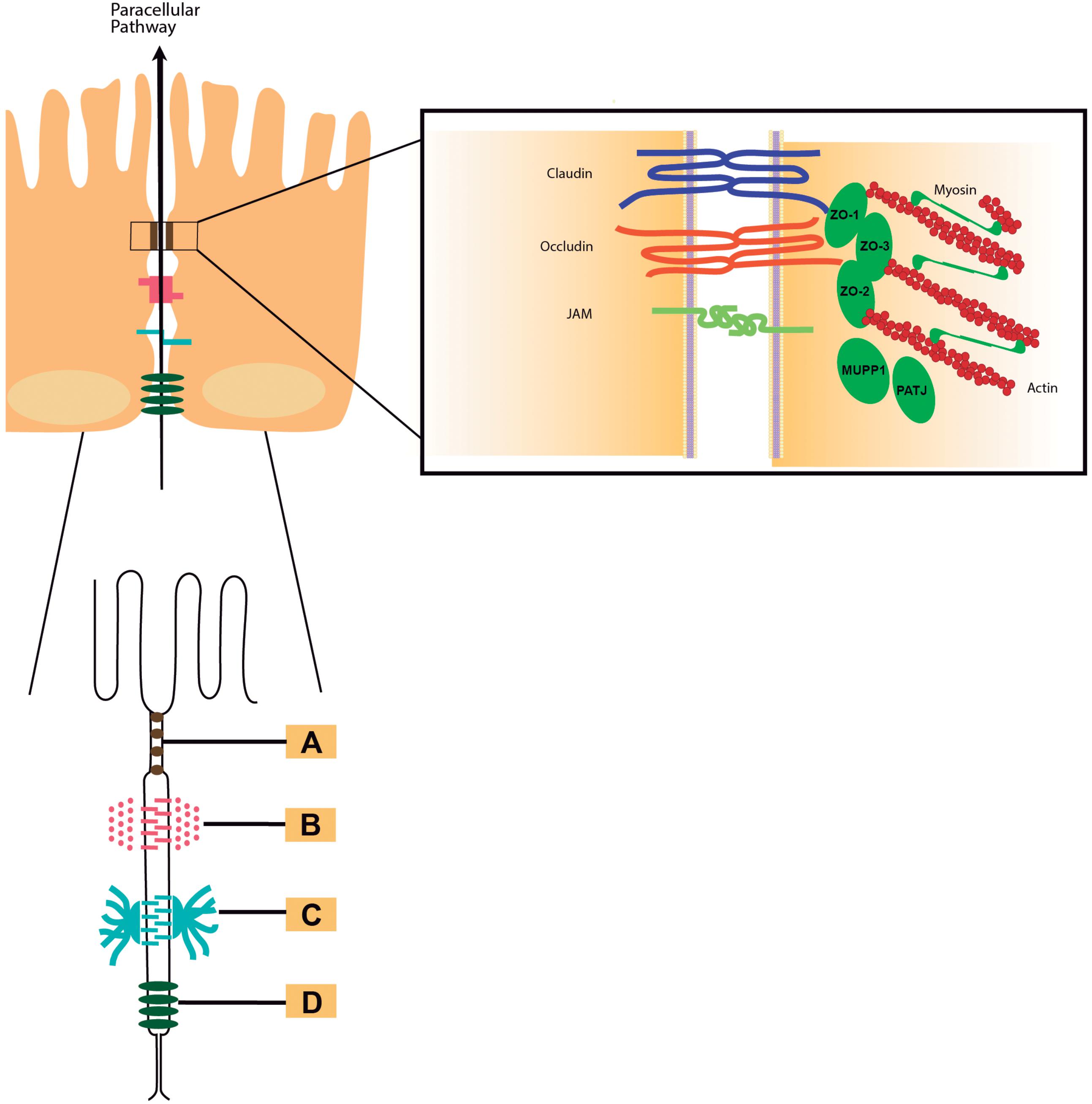
Frontiers | Accessory Toxins of Vibrio Pathogens and Their Role in Epithelial Disruption During Infection

Continuous Assembly of β-Roll Structures Is Implicated in the Type I-Dependent Secretion of Large Repeat-in-Toxins (RTX) Proteins - ScienceDirect

Channel formation by RTX-toxins of pathogenic bacteria: Basis of their biological activity - ScienceDirect

Templated folding of the RTX domain of the bacterial toxin adenylate cyclase revealed by single molecule force spectroscopy | Nature Communications

The Vibrio cholerae MARTX toxin simultaneously induces actin collapse while silencing the inflammatory response to cytoskeletal damage | bioRxiv

Vibrio cholerae Strains with Mutations in an Atypical Type I Secretion System Accumulate RTX Toxin Intracellularly | Journal of Bacteriology