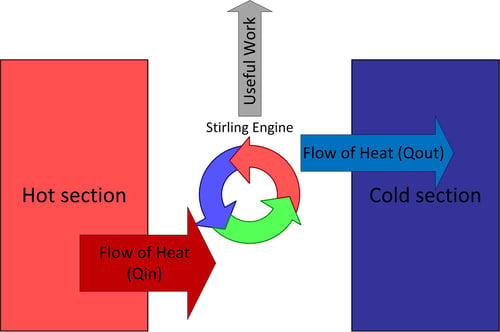
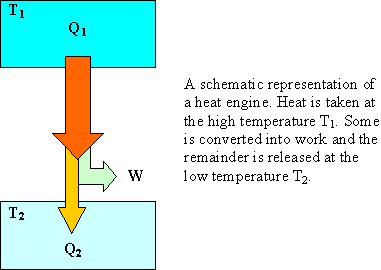
Calculate the difference between the two specific heats of Helium gas per gram. Given that molecular weight of He = 4, J = 4.18 joule/cal and R = 8.31 J mole^-1//K^-1?
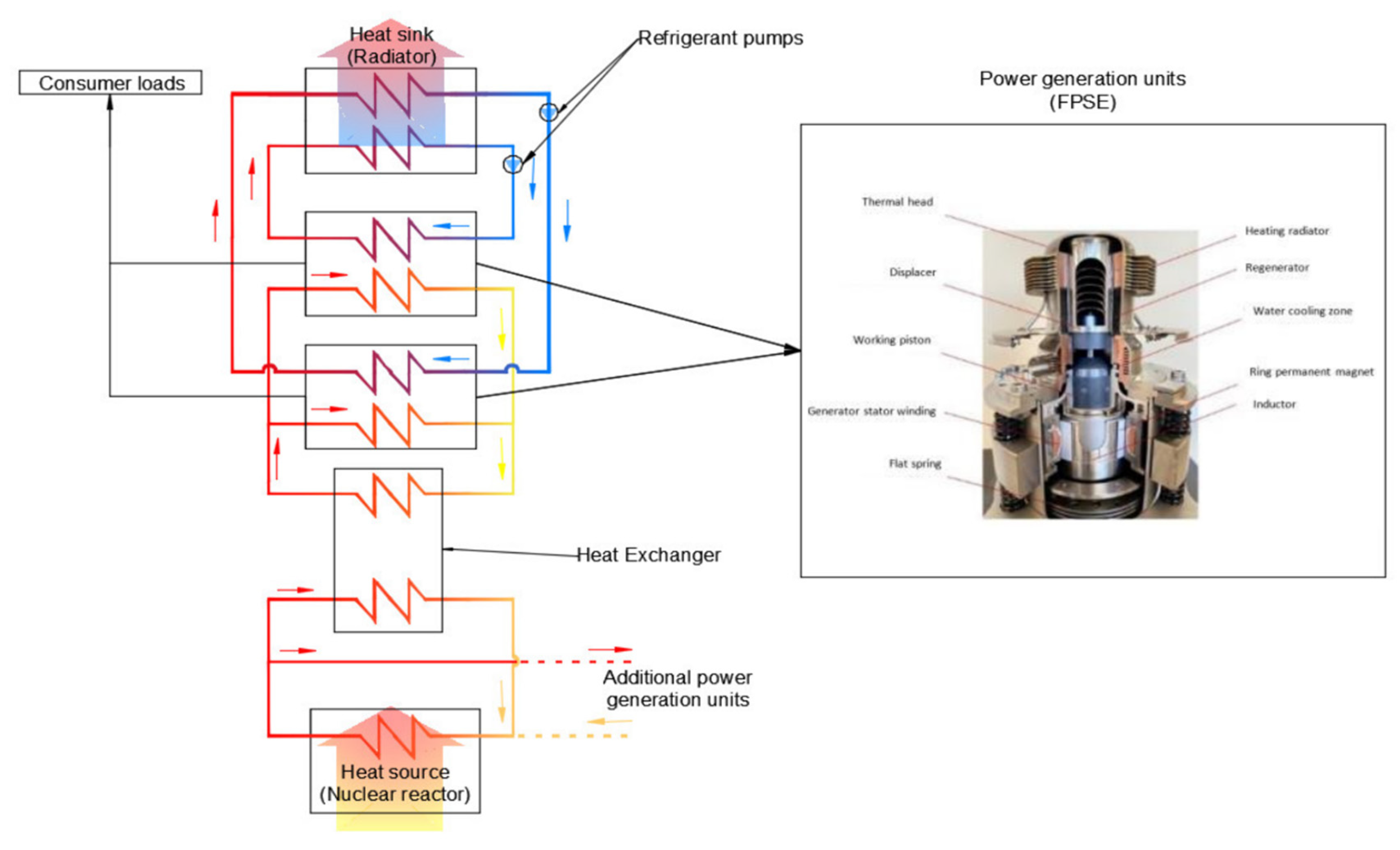
Symmetry | Free Full-Text | An Enhanced Calculation Method of the Heat Rejection System of a Free-Piston Stirling Engine (FPSE) Operating on the Moon

The operation of a certain heat engine takes an ideal monatomic gas through a cycle shown as the rectangle on the PV diagram below. a) Determine the efficiency of this engine. Let
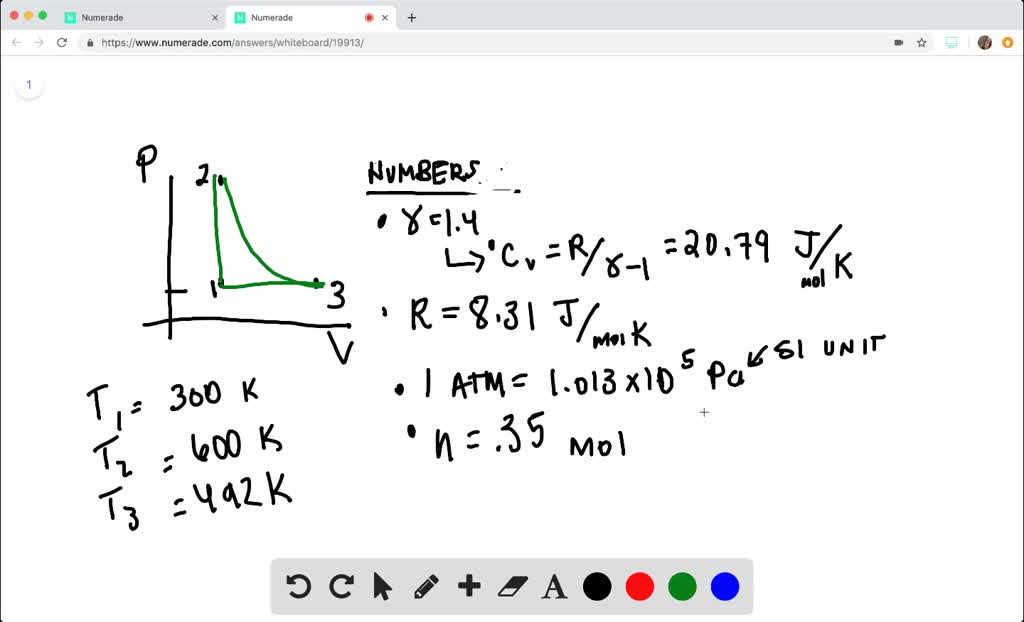
SOLVED:A heat engine takes 0.350 mol of a diatomic ideal gas around the cycle shown in the pV-diagram of Fig. P20.36. Process 1→2 is at constant volume, process 2→3 is adiabatic, and
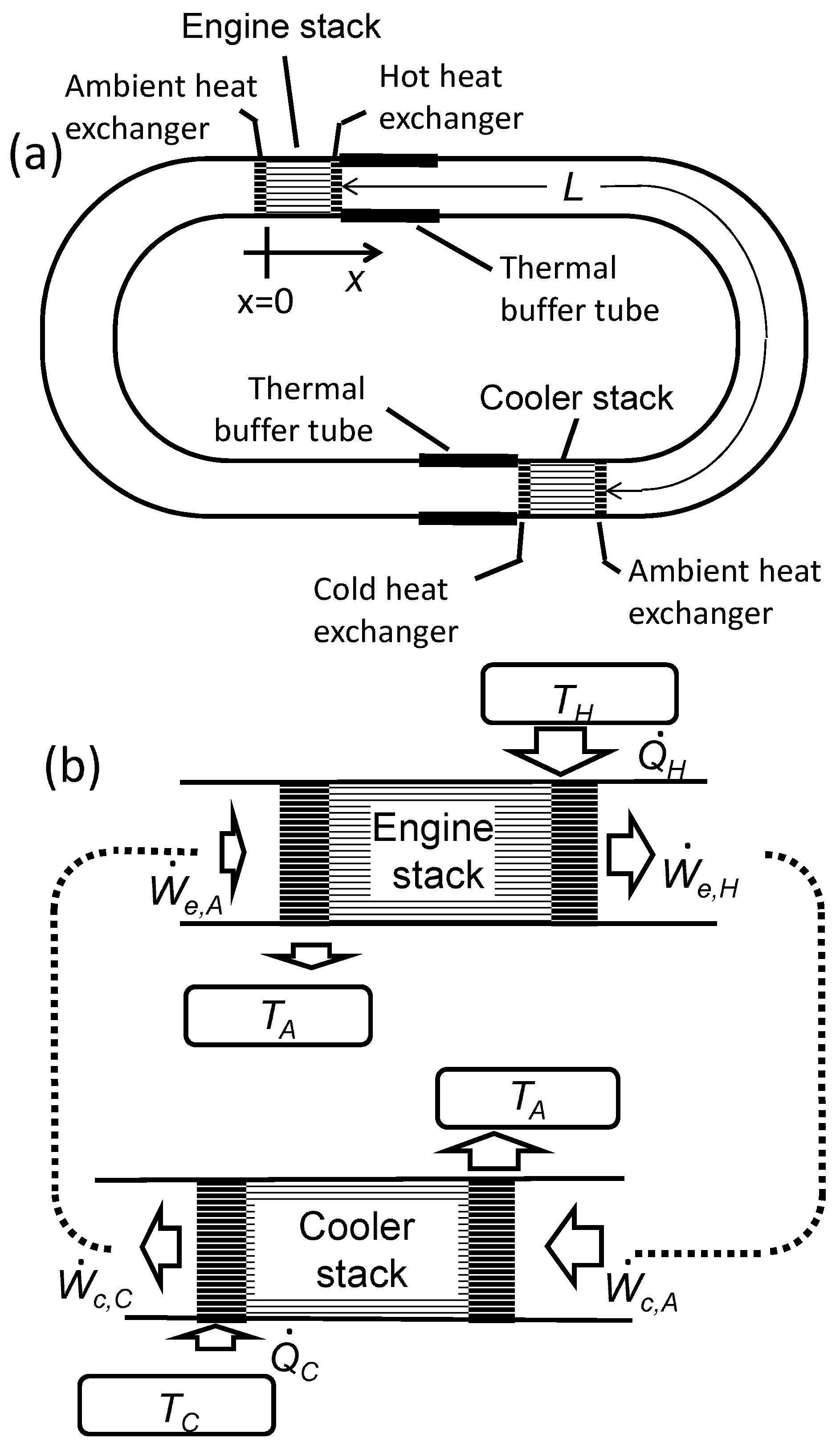
Applied Sciences | Free Full-Text | Numerical Calculation of the Performance of a Thermoacoustic System with Engine and Cooler Stacks in a Looped Tube